mRNA iPSC
New Allele Biotech Publication on Stem Cells
Feeder-Free Reprogramming of Human Fibroblasts with Messenger RNA
Current Protocols in Stem Cell Biology • November 13, 2013
DOI: 10.1002/9780470151808.sc04a06s27
Authors: Luigi Warren, Jiwu Wang
This unit describes a feeder-free protocol for deriving induced pluripotent stem cells (iPSCs) from human fibroblasts by transfection of synthetic mRNA. The reprogramming of somatic cells requires transient expression of a set of transcription factors that collectively activate an endogenous gene regulatory network specifying the pluripotent phenotype. The necessary ectopic factor expression was first effected using retroviruses; however, as viral integration into the genome is problematic for cell therapy applications, the use of footprint-free vectors such as mRNA is increasingly preferred. Strong points of the mRNA approach include high efficiency, rapid kinetics, and obviation of a clean-up phase to purge the vector. Still, the method is relatively laborious and has, up to now, involved the use of feeder cells, which brings drawbacks including poor applicability to clinically oriented iPSC derivation. Using the methods described here, mRNA reprogramming can be performed without feeders at much-reduced labor and material costs relative to established protocols.
Allele iPSC Service and Technology Licensing Contact: http://www.allelebiotech.com/cell-line-and-culture-services/#ips-line
New Allele Product of the Month: FP-nAb™ products for 100% pull-down
Picture Blog: mRNA Reprogramming for Human iPSCs without B18R!
Human induced pluripotent stem cells provide a great route towards personalized medicine and high accuracy drug screening. Allele Biotech has developed the most efficient method of making human iPSCs by using enhanced mRNAs, which have been adopted by leading pharmaceutical companies for clinical trials. The effects from medium-supplementing mRNAs are robust yet transient, and highly specific compared to both miRNAs (off-targets) and small molecules (unknown targets). To repress cellular immune response to introduced RNA molecules, viral protein B18R was previously used during mRNA reprogramming.
B18R is relatively expensive and inconvenient to use because it requires pre-aliquoting and -80C storage. The protocol has recently been dramatically improved at Allele through an NIDA-funded project. In our latest reprogramming run, all we needed to do was to include mRNA complex in the supplement during medium change for just a week without the need of adding any other type of molecules (such as B18R, miRNA, or chemicals) to help the mRNA mix, unlike all other known mRNA-reprogramming protocols. This advancement can make reprogramming human fibroblasts to footprint-free and xeno-free iPSCs a routine experiment for any lab to perform.
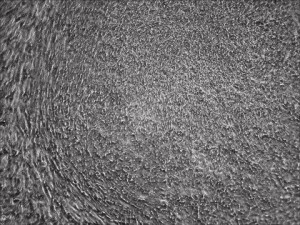
Human R-iPSCs were created without the need of B18R, dramatically reduced the cost and inconvenience. Shown is a newly formed iPSC colony.
Allele Biotech Receives $200,000 Grant to Update Its mRNA Reprogramming Commercial Products and Services
On June 10, 2013 Allele received an SBIR award from the National Institute of Drug Abuse (NIDA/NIH) entitled “Revolutionary Technology for Efficient Derivation of Human iPSCs with Messenger RNA”. The goal of the proposed project is to provide to the biomedical research market an advanced reagent kit and services for highly efficient reprogramming of high quality human induced pluripotent stem cells (iPSCs). At the core of this kit is the Allele team’s recent development transcribed messenger RNA (mRNA). Compared to other reprogramming methods, such as lentivirus, Sendai virus, protein, small molecules or any combinations of these reagents, our new generation of the mRNA method often requires less than half the time while sometimes achieving “bulk conversion” efficiency.
While the Allele reprogramming technology was designed for clinical use as the process is feeder-free, xeno-free, chromosome integration-free, as well as without the need for cell splitting, PI, Dr. Jiwu Wang states, “Our purpose of executing the NIH-funded research it to make our method so easy that any researcher can integrate iPSC into his or her projects.” In addition to the extremely high efficiency, mRNA-generated iPSCs should also be more stable because there are no genetic alterations, more uniform among all clones as there is no clonal event, and ultimately suitable for future autologous cell therapy now that creating iPSCs from patient tissue cells should no longer be the rate-limiting steps.
Allele’s business model is to provide cGMP-grade iPSCs to pharmaceutical companies and perform large scale reprogramming by partnering first with university-affiliated hospitals. Great progress has been made in both directions, which has prompted the initiation of a cGMP unit within Allele’s newly acquired building in San Diego.
mRNA Transfection for Better Transgene Expression
Different approaches have been developed to over-express or ectopically express a protein in cells: peptide or full length recombinant protein transfer, viral gene transfer, non-viral DNA transfer and non-viral mRNA transfer.
1) Peptide transfection can be efficient, yet it is limited to only a small part of the protein, limiting the functional potential. Protein transfection is not consistent enough so far, because of the complicated properties of different proteins. Allele Biotech has tested dozens of proteins with several proprietary reagents, leader peptides, etc. but we have decided not to carry a protein transfection product line due to its instability. Furthermore, protein production is an expensive and laborious process.
2) Viral gene transfer is very effective, such as the HIV-based lentivirus or MMLV-based retrovirus, adenovirus, adeno-like virus or baculovirus, etc. However, the potent side-effect will still need to be considered for certain applications, especially involving clinical studies. Nevertheless, as research tools, viral gene transfer is still a highly preferred method. Allele Biotech has been providing the most effective platform for both MMLV-based and HIV-1-based retrovirus packaging. Check out our product website for details.
3) Non-viral DNA transfer is the most widely used transgene method in the biological research community, due to the simplicity of the procedure. There are many commercial kits on the market. However, the low efficiency for transfecting most primary cells significantly limits their use. In recent years, several leading biotech companies have developed various electroporation systems to improve the transfection efficiency and cell viability; although these improvements help with getting DNA inside the cytoplasm, they hardly help transport it into nucleus where DNA is transcribed.
4) Non-viral mRNA transfer has been around for a long time, but it is not widely used. It made a big splash recently through its use for iPSCs reprogramming. IPSCs factor mRNAs greatly improved the iPSCs induction efficiency and completely avoided the viral integration. Other well-known examples of mRNA transfection include loading special cancer antigens or HIV antigens to dendritic cells (DCs) in vitro for personal immunotherapy. PSA antigen expressing DCs transfected by mRNA has moved on to Phrase I Clinical Trials for this purpose.
New Product of the Week: 3C protease immobilized on beads for GST, His tag removal, email oligo@allelebiotech.com for details.
Promotion of the week: 10% off on all fluorescent proteins. To redeem, email oligo@allelebiotech.com along with PROMO code: JELLYFISH
Categories
- Allele Mail Bag
- cGMP
- Customer Feedback
- Fluorescent proteins
- iPSCs and other stem cells
- nAb: Camelid Antibodies, Nanobodies, VHH
- Next Generation Sequencing (NextGen Seq)
- NIH Budget and You
- oligos and cloning
- Open Forum
- RNAi patent landscape
- SBIR and Business issues
- State of Research
- Synthetic biology
- Uncategorized
- Viruses and cells
- You have the power
Archives
- October 2018
- April 2018
- March 2018
- January 2018
- October 2017
- September 2017
- August 2017
- March 2017
- February 2017
- January 2017
- November 2016
- September 2016
- August 2016
- July 2016
- June 2016
- May 2016
- April 2016
- February 2016
- October 2015
- September 2015
- August 2015
- June 2015
- March 2015
- January 2015
- December 2014
- March 2014
- February 2014
- January 2014
- December 2013
- November 2013
- October 2013
- September 2013
- August 2013
- July 2013
- June 2013
- May 2013
- April 2013
- March 2013
- January 2013
- December 2012
- November 2012
- October 2012
- September 2012
- August 2012
- July 2012
- May 2012
- April 2012
- February 2012
- January 2012
- December 2011
- November 2011
- October 2011
- September 2011
- August 2011
- July 2011
- June 2011
- May 2011
- April 2011
- March 2011
- February 2011
- January 2011
- December 2010
- November 2010
- October 2010
- September 2010
- August 2010
- July 2010
- June 2010
- May 2010
- April 2010
- March 2010
- February 2010
- January 2010
- December 2009
- November 2009
- October 2009
- September 2009
- August 2009
- July 2009
- June 2009
- May 2009
- April 2009
- March 2009
- February 2009
- January 2009
- December 2008
- October 2008
- August 2008
- July 2008



