mRNA transfection
mRNA Delivery And the Next Wave of Regenerative Medicine
Published online by Nature Biotechnology, researchers from Ken Chien’s lab at Harvard and other coauthors showed that modified mRNA of VEGF-A injected intramyocardially resulted in the expansion and directed differentiation of endogenous heart progenitors. VEGF-A modRNA markedly improved heart function and enhanced long-term survival of recipients by directing epicardial progenitor cells toward cardiovascular cell types. This publication appears to be the first example of using mRNA as a delivery platform for cell fate-related therapy. AstraZeneca recently invested $240 million on mRNA-related delivery via Moderna, a company with roots within the Harvard stem cell group.
The drastically increased efficacy of using the mRNA platform was accredited to the pulse-like kinetics of mRNA expression profile. It was explained by the fact that native paracrine signals are often transient and precisely regulated in time and space, therefore the pulse-like expression profile of modRNA might be well suited to delivering paracrine-factor signals. Transfected mRNA molecules do not need to penetrate the nuclear membrane, which greatly enhances the efficiency of protein expression on a per transfected molecule over DNA. mRNAs turn over in a much faster pace than plasmid-mediated transgene expression. This is beneficial to many cell fate decisions as exemplified by this recent publication.
Allele Biotech’s reprogramming technologies, licensed by some of the leading stem cell therapy companies, are built around the mRNA platform. We chose mRNA as our core technology to not only change cell fate, but also direct differentiation. We know this platform is the future for cell fate manipulation because we have seen how robustly mRNA expression made the day-and-night difference in gene expression when compared to plasmid DNA (episomal or not), retrovirus, lentivirus, baculo virus, or even transfected proteins. We could convert human fibroblasts into iPSCs, in bulk, in as short as one week with no more effort than changing mRNA complex-containing medium.
Another recent development in iPSC research is in situ reprogramming. Abad et al. generated mice carrying a Tet-inducible cassette of the four cell-reprogramming factors. They then added feed doxycycline to the animals. After several weeks, teratomas appeared in various tissues, indicating that in situ reprogramming had occurred. The iPSCs created this way did not appear to have much advantage over in vitro produced iPSCs other than they are totipotent (helpful if you are studying placenta). Nevertheless, the concept of changing cell fate in situ as dramatically as complete reprogramming is an important leap of faith. As for the next big step, it is easy to see that mRNAs are well suited for in situ reprogramming, as well as transdifferentiation, and more complex gene delivery than the above mentioned VEGF-A alone in heart treatment.
References:
Zangi et al. Nature Biotechnology, http://www.nature.com/nbt/journal/vaop/ncurrent/abs/nbt.2682.html
Abad et al. Nature, http://www.nature.com/nature/journal/vaop/ncurrent/abs/nature12586.html
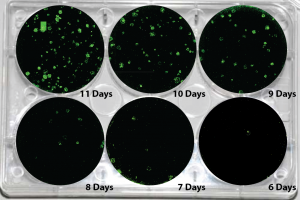
Picture Blog — Human mRNA-Induced Pluripotent Stem Cells Generated in Days
R-iPS Cell FAQ 1:
What phenotypic changes can be observed during a successful reprogramming trial?
About one week out, target fibroblasts should show an involution of fibroblastic processes, and foci or clusters of epitheliod cells—ideally with small nuclei, minimal cytoplasm, and signs of ongoing mitosis—should appear. Colonies with hESC morphology typically start emerging in ~10-14 days.
Methods of iPSC Generation Update
Induced pluripotent stem cells can be directly generated from adult cell cultures through the introduction of a group of factors, e.g. Oct4, Sox2, Klf4, and c-Myc (the Yamanaka factors) [Takahashi and Yamanaka, 2006]. Additional factors such as Nanog and Lin28 can either substitute some of the Yamanaka factors or supplement them for higher reprogramming efficiency [Yu et al. 2007].
The original pluripotent stem cells induction methods involved retrovirus or lentivirus that would leave foot-print in the host genome, a concern for clinical use of iPSCs. Several groups have tried to create iPSCs without integrating viruses, such as using small molecules, directly delivering proteins instead of cDNAs, viruses with RNA genomes, episomal systems, or removable elements such as PiggyBac or Sleeping Beauty transposons. From the literature and our first-hand experience in the iPS market, none of these methods has become a widely applicable tool, mostly due to impractically low reprogramming efficiency.
In addition to low efficiency, RNA viruses, such as the sendai virus, are still viruses and have virus-associated risks. Episomal plasmids or removable transposons still involve DNA, so the possibility of genomic integration by recombination remains. In case of some transposons such as PiggyBac, there is an additional question about the degree of removal – whether it is certain that all integrated transposons, often inserted within genes, are deleted; in case of transposons similar to Sleeping Beauty, the small footprints they leave behind may post a concern.
The method of choice for generating zero-footprint iPSCs should clearly be RNA-based without the involvement of virus. Luigi Warren and his former colleagues at Harvard demonstrated that by using in vitro transcribed iPS factor mRNAs with modified CTP and UTP, and 5’-cap can effectively reprogram a number of different human as well as mouse cells. The efficiency even exceeds those by using retrovirus or lentivirus by 10 to 100 fold. Furthermore, the RiPSCs created with mRNAs appear to be closer to hESCs as shown by expression profiling.
Very recently, a few miRNAs that have high expression levels in stem cells were shown to be able to reprogram mouse and human somatic cells when expressed together from a lentivirus [Anokye-Danso et al. 2011]. while that work used lentivirus, thus not directly applicable to the current project, Miyoshi et al. later showed that by using synthesized mature miRNA (overlapping but not the same set of miRNAs as used by Anokye-Danso et al.) reprogramming cold be achieved without viral infection. We believe that this is a promising method and would like to pursue it further and to find out whether these mi-iPSCs relate to hESCs as closely as R-iPSCs. Because transfecting synthetic miRNAs “does operate at considerably lower efficiency” in terms of iPSC creation [Miyoshi et al. 2011],alternative protocols may include transfecting the iPS factor mRNAs together with various miRNAs at different doses and frequencies.
New Product of the Week: EF1a-lacZ lentivirus particles, for expressing nuclear lacZ in virtually any human or mouse cells.
This week save 25% on photoconvertible fluorescent protein mClavGR2 cloning plasmids. Email oligo@allelebiotech.com with code FPBLOG0831.
Blog References: Warren, L. et al. “Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA” 2010 Cell Stem Cell 7(5): 618-30
Anokye-Danso, F. et al. “Highly efficient miRNA-mediated reprogramming of mouse and human somatic cells to pluripotency” 2011 Cell Stem Cell 8(4): 376-88
Miyoshi, N. et al. “Reprogramming of mouse and human cells to pluripotency using mature microRNAs” 2011 Cell Stem Cell 8(6): 633-8
Kim, H. et al. “miR-371-3 expression predicts neural differentiation propensity in human pluripotent stem cells” 2011 Cell Stem Cell 8(6): 695-706
Takahashi, K. and Yamanaka, S. “Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors” 2006 Cell 126(4): 663-76
Yu, J. et al. “Induced pluripotent stem cell lines derived from human somatic cells” 2007 Science 318(5858): 1917-20
mRNA Transfection for Better Transgene Expression
Different approaches have been developed to over-express or ectopically express a protein in cells: peptide or full length recombinant protein transfer, viral gene transfer, non-viral DNA transfer and non-viral mRNA transfer.
1) Peptide transfection can be efficient, yet it is limited to only a small part of the protein, limiting the functional potential. Protein transfection is not consistent enough so far, because of the complicated properties of different proteins. Allele Biotech has tested dozens of proteins with several proprietary reagents, leader peptides, etc. but we have decided not to carry a protein transfection product line due to its instability. Furthermore, protein production is an expensive and laborious process.
2) Viral gene transfer is very effective, such as the HIV-based lentivirus or MMLV-based retrovirus, adenovirus, adeno-like virus or baculovirus, etc. However, the potent side-effect will still need to be considered for certain applications, especially involving clinical studies. Nevertheless, as research tools, viral gene transfer is still a highly preferred method. Allele Biotech has been providing the most effective platform for both MMLV-based and HIV-1-based retrovirus packaging. Check out our product website for details.
3) Non-viral DNA transfer is the most widely used transgene method in the biological research community, due to the simplicity of the procedure. There are many commercial kits on the market. However, the low efficiency for transfecting most primary cells significantly limits their use. In recent years, several leading biotech companies have developed various electroporation systems to improve the transfection efficiency and cell viability; although these improvements help with getting DNA inside the cytoplasm, they hardly help transport it into nucleus where DNA is transcribed.
4) Non-viral mRNA transfer has been around for a long time, but it is not widely used. It made a big splash recently through its use for iPSCs reprogramming. IPSCs factor mRNAs greatly improved the iPSCs induction efficiency and completely avoided the viral integration. Other well-known examples of mRNA transfection include loading special cancer antigens or HIV antigens to dendritic cells (DCs) in vitro for personal immunotherapy. PSA antigen expressing DCs transfected by mRNA has moved on to Phrase I Clinical Trials for this purpose.
New Product of the Week: 3C protease immobilized on beads for GST, His tag removal, email oligo@allelebiotech.com for details.
Promotion of the week: 10% off on all fluorescent proteins. To redeem, email oligo@allelebiotech.com along with PROMO code: JELLYFISH
Expression of iPS Factors from Transfected mRNA
Differentiated cells can be reprogrammed to pluripotency by enforced expression of certain combinations of stem cell-specific protein factors in them. The power of this method was first demonstrated by Yamanaka’s group using retroviruses carrying Oct3/4, Sox2, c-Myc, and Klf4. Alternative factors such as Lin28 and Nanog, and additional factors such as the human telomerase gene hTert and shRNA against p53 were also shown to contribute to reprogramming. From the very beginning it was realized that viral integration would pose a major problem in using the induced pluripotent stem cells (iPSCs) for clinical purposes. There have been multiple attempts to circumvent this problem by using non-integrating vectors such as plasmid, minicircle DNA, adenovirus, baculovirus, removable transposons, episomal DNA, or by introducing recombinant proteins with a transmembrane domain into target cells. From reports in the field and customer feedbacks it seems that retroviral or lentiviral systems are still the most efficient in reprogramming. mRNA is about the only option left unreported, until an article by Warren et al was published in Cell Stem Cell online recently.
From that report, it is clear that the reason that it took so long for RNA-induced iPSCs (RiPSCs) to appear in the literature was because synthetic mRNAs activate interferon responses in mammalian cells, reminding us of the early days of RNAi. The authors took a number of steps to reduce interferon responses, including adding a 5’-cap (actually a fairly standard step in in vitro transcription), using a phosphatase to remove 5’ triphosphates on uncapped mRNAs, and using modified C and U bases (5-methucytidine or 5mC and pseudouridine or psi) during T7 promoter-driven in vitro transcription. The prepared mRNA was then administered everyday for 17 days at an amount not clearly defined in the paper. The main benefit of this method is of course that there is no gene integration to alter the chromosome. The efficiency of the new method was also compared to using viral vectors and it was shown that 1.4% conversion efficiency was achieved vs retroviral systems’ 0.01% (although we have experienced better results using lentivirus, at least the 4-in-1 version).
The DNA templates used for in vitro transcription of the iPS factors were created by multiple PCR reactions and bridged ligation; it could also be done by other cloning strategies. For those excited about trying this new way of making iPSCs, the major hassle would be preparing modified mRNAs good and abundant enough for 17 consecutive transfections. Allele Biotech would like to provide custom services, before offering shelf products, for creating such mRNAs as the method sounds potentially very helpful to many researchers in the iPSC field.
- New Product of the Week 100410-101010:
pLICO-mWasabi (Promoterless FP Reporter Vector ), listed as product-on-demand, now available, ABP-HL-PE40010 $395.00.
- Promotion of the Week 100410-101010:
Barrier too high to start using virus? Allele lowers it for starters, $500 for bactulo virus protein production, and $300 retrovirus packaging. Code 100310VIVEC, email vivec@allelebiotech.com
Categories
- Allele Mail Bag
- cGMP
- Customer Feedback
- Fluorescent proteins
- iPSCs and other stem cells
- nAb: Camelid Antibodies, Nanobodies, VHH
- Next Generation Sequencing (NextGen Seq)
- NIH Budget and You
- oligos and cloning
- Open Forum
- RNAi patent landscape
- SBIR and Business issues
- State of Research
- Synthetic biology
- Uncategorized
- Viruses and cells
- You have the power
Archives
- October 2018
- April 2018
- March 2018
- January 2018
- October 2017
- September 2017
- August 2017
- March 2017
- February 2017
- January 2017
- November 2016
- September 2016
- August 2016
- July 2016
- June 2016
- May 2016
- April 2016
- February 2016
- October 2015
- September 2015
- August 2015
- June 2015
- March 2015
- January 2015
- December 2014
- March 2014
- February 2014
- January 2014
- December 2013
- November 2013
- October 2013
- September 2013
- August 2013
- July 2013
- June 2013
- May 2013
- April 2013
- March 2013
- January 2013
- December 2012
- November 2012
- October 2012
- September 2012
- August 2012
- July 2012
- May 2012
- April 2012
- February 2012
- January 2012
- December 2011
- November 2011
- October 2011
- September 2011
- August 2011
- July 2011
- June 2011
- May 2011
- April 2011
- March 2011
- February 2011
- January 2011
- December 2010
- November 2010
- October 2010
- September 2010
- August 2010
- July 2010
- June 2010
- May 2010
- April 2010
- March 2010
- February 2010
- January 2010
- December 2009
- November 2009
- October 2009
- September 2009
- August 2009
- July 2009
- June 2009
- May 2009
- April 2009
- March 2009
- February 2009
- January 2009
- December 2008
- October 2008
- August 2008
- July 2008